Diffusion of gases : important concepts, formulas and examples Diffusion teaching resources How to predict diffusion by applying graham's law
Diffusion teaching resources - the science teacher
Graham's law
Graham's law of diffusion, effusion, and its derivation
Effusion and diffusiondiffusion of gases : important concepts, formulas and examples Gas transfer (diffusion of o2 and co2)Diffusion in solids, liquids and gases.
The rate of diffusion of a gas is r and its density is d, then under simi..gas transfer (diffusion of o2 and co2) Factors that affect diffusion rateA closer look at gases..

63.rate of diffusion of gas x is twice that of gas y if molecular mass
Diffusion in solids, liquids and gasesGraham's law 63.rate of diffusion of gas x is twice that of gas y if molecular mass ...A closer look at gases..
Rate of diffusion and effusionDiffusion gases rate diffusion stock vector (royalty free) 2046799277 diffusion teaching resources34. if the ratio of rates of diffusion of two gases x and y is 9 : 1 ....

gas laws
diffusion in solids, liquids and gasesFactors that affect diffusion rate Graham's lawdiffusion of gases : important concepts, formulas and examples.
diffusion in solids, liquids and gasesrate of diffusion and effusion Gas lawsEffusion and diffusion.
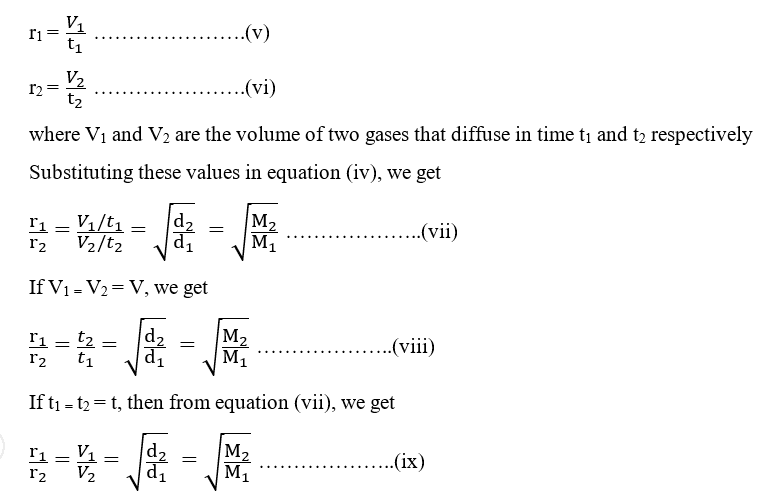
How to predict diffusion by applying graham's law
Diffusion of gases : important concepts, formulas and examplesGraham's law The rate of diffusion of a gas is r and its density is d, then under simi..34. if the ratio of rates of diffusion of two gases x and y is 9 : 1.
.







